
Marvin eTMF
Marvin eTMF is a compliant, validated, and budget-friendly eClinical solution for managing all your essential trial documentation.
What is Marvin eTMF?
Marvin eTMF supports better project management through real-time access to study documentation. It offers clear workflows, reporting, search and retrieval, audit-readiness at each point in time, and many other valuable features. Marvin eTMF is a powerful and intuitive system answering our clients needs for managing documentation in a secure and regulatory compliant manner. It is a cloud-based, highly configurable eTMF system with quality-focused workflows and metada recording.
Screenshots
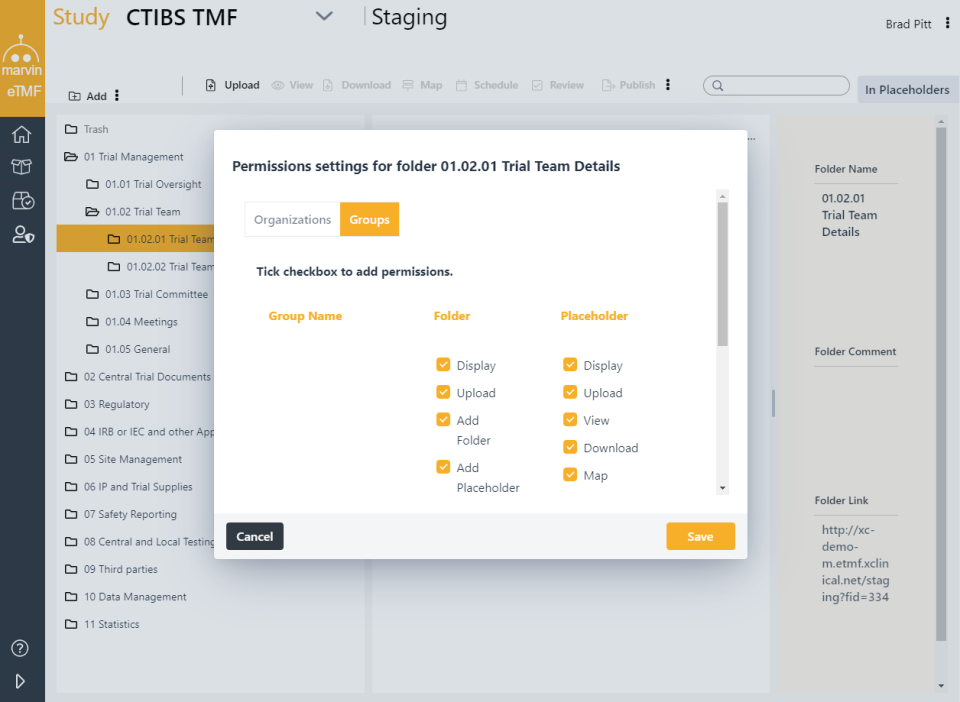

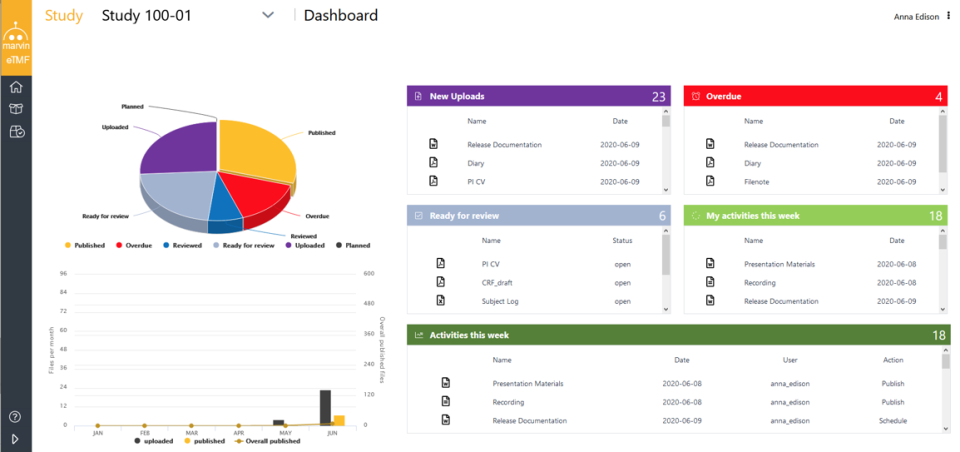
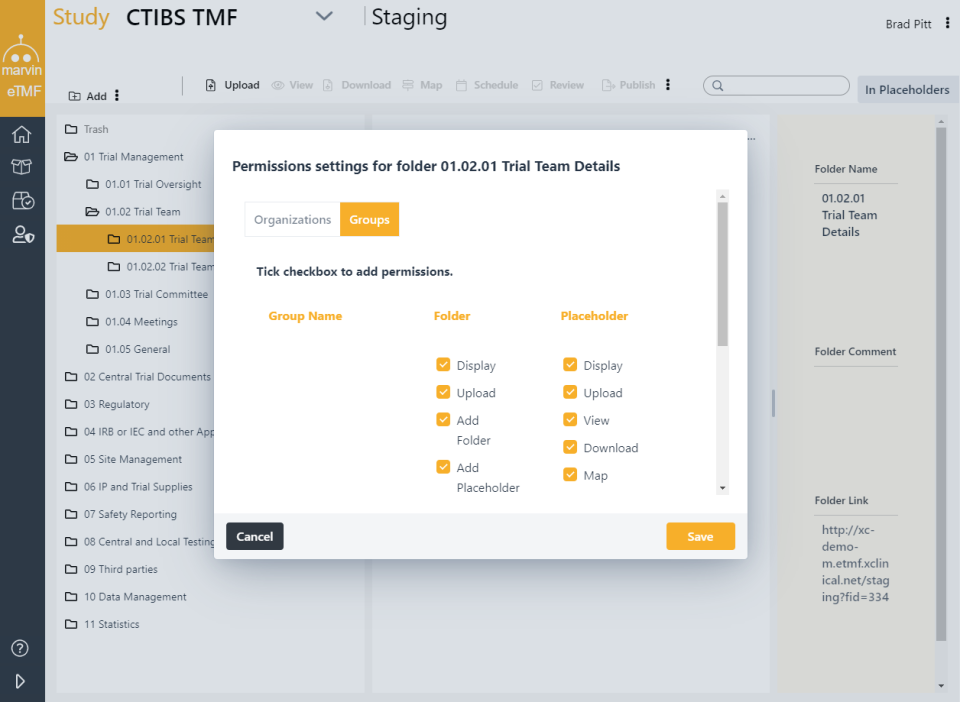
Categories
Features
21 CFR Part 11 Compliance
Audit Trail
Data Import/Export
Document Management
Electronic Data Capture
HIPAA Compliant
Workflow Management
FAQ
The official website of Marvin eTMF is https://www.evidentiq.com/
Marvin eTMF supports better project management through real-time access to study documentation. It offers clear workflows, reporting, search and retrieval, audit-readiness at each point in time, and many other valuable features. Marvin eTMF is a powerful and intuitive system answering our clients needs for managing documentation in a secure and regulatory compliant manner. It is a cloud-based, highly configurable eTMF system with quality-focused workflows and metada recording.
Marvin eTMF belongs to the Clinical Trial Management category.
Marvin eTMF offers features such as 21 CFR Part 11 Compliance, Audit Trail, Data Import/Export, Document Management, Electronic Data Capture, HIPAA Compliant, Workflow Management.
No, Marvin eTMF does not offer a free trial.
Pricing
Starting at:
$455/per month
Free Trial Available