
RegDocs365
Regulated Content and Collaboration Solutions for Life Sciences
What is RegDocs365?
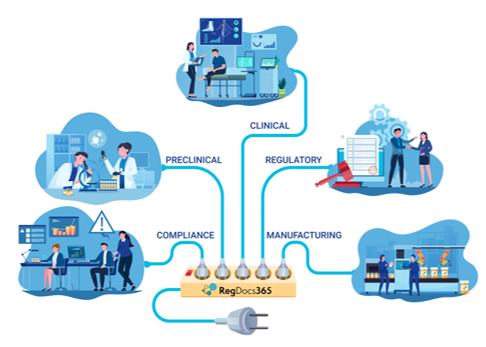
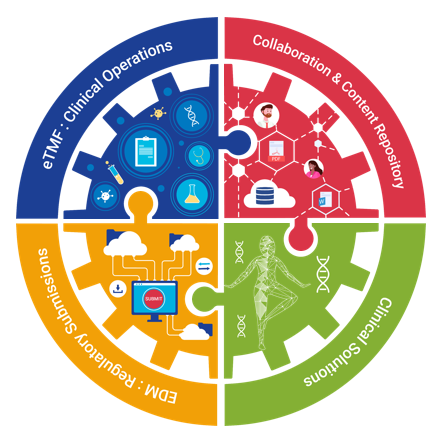
Regulated Content and Collaboration Solutions for Life Sciences RegDocs365 is a fully 21 CFR Part 11 compliant, ready-to-use, cost-efficient content and document management solution hosted in an Audit Ready Compliant Cloud (ARCC) validated environment. As a turn-key content management solution built specifically for Life Science companies, RegDocs365 supports life science companies actively bringing drugs, biologics, or devices to market in their efforts to prepare regulatory submissions. RegDocs365 provides a Life Science focused platform for all of your workflow, dashboard and document management needs in a fully regulatory compliant fashion. Easily streamline workflows at any stage of development through commercialization, including discovery, clinical trials, regulatory submissions, and post-market. RegDocs365 effectively centralizes and manages all digital content (documents, data, voice, and video) to create, protect, and publish in multi-sponsor environments.
Screenshots



Video
Categories
Features
21 CFR Part 11 Compliance
Audit Trail
Data Capture and Transfer
Data Import/Export
Document Management
Electronic Data Capture
HIPAA Compliant
Workflow Management
FAQ
The official website of RegDocs365 is https://www.courtsquaregroup.com/
Regulated Content and Collaboration Solutions for Life Sciences RegDocs365 is a fully 21 CFR Part 11 compliant, ready-to-use, cost-efficient content and document management solution hosted in an Audit Ready Compliant Cloud (ARCC) validated environment. As a turn-key content management solution built specifically for Life Science companies, RegDocs365 supports life science companies actively bringing drugs, biologics, or devices to market in their efforts to prepare regulatory submissions. RegDocs365 provides a Life Science focused platform for all of your workflow, dashboard and document management needs in a fully regulatory compliant fashion. Easily streamline workflows at any stage of development through commercialization, including discovery, clinical trials, regulatory submissions, and post-market. RegDocs365 effectively centralizes and manages all digital content (documents, data, voice, and video) to create, protect, and publish in multi-sponsor environments.
RegDocs365 belongs to the Clinical Trial Management category.
RegDocs365 offers features such as 21 CFR Part 11 Compliance, Audit Trail, Data Capture and Transfer, Data Import/Export, Document Management, Electronic Data Capture, HIPAA Compliant, Workflow Management.
No, RegDocs365 does not offer a free trial.
Pricing
Starting at:
$100/per month
Free Trial Available