
ShareCRF
Are your study data working for you, or are you working for them? Control, compliance, and adaptability shouldn be a struggle.
What is ShareCRF?
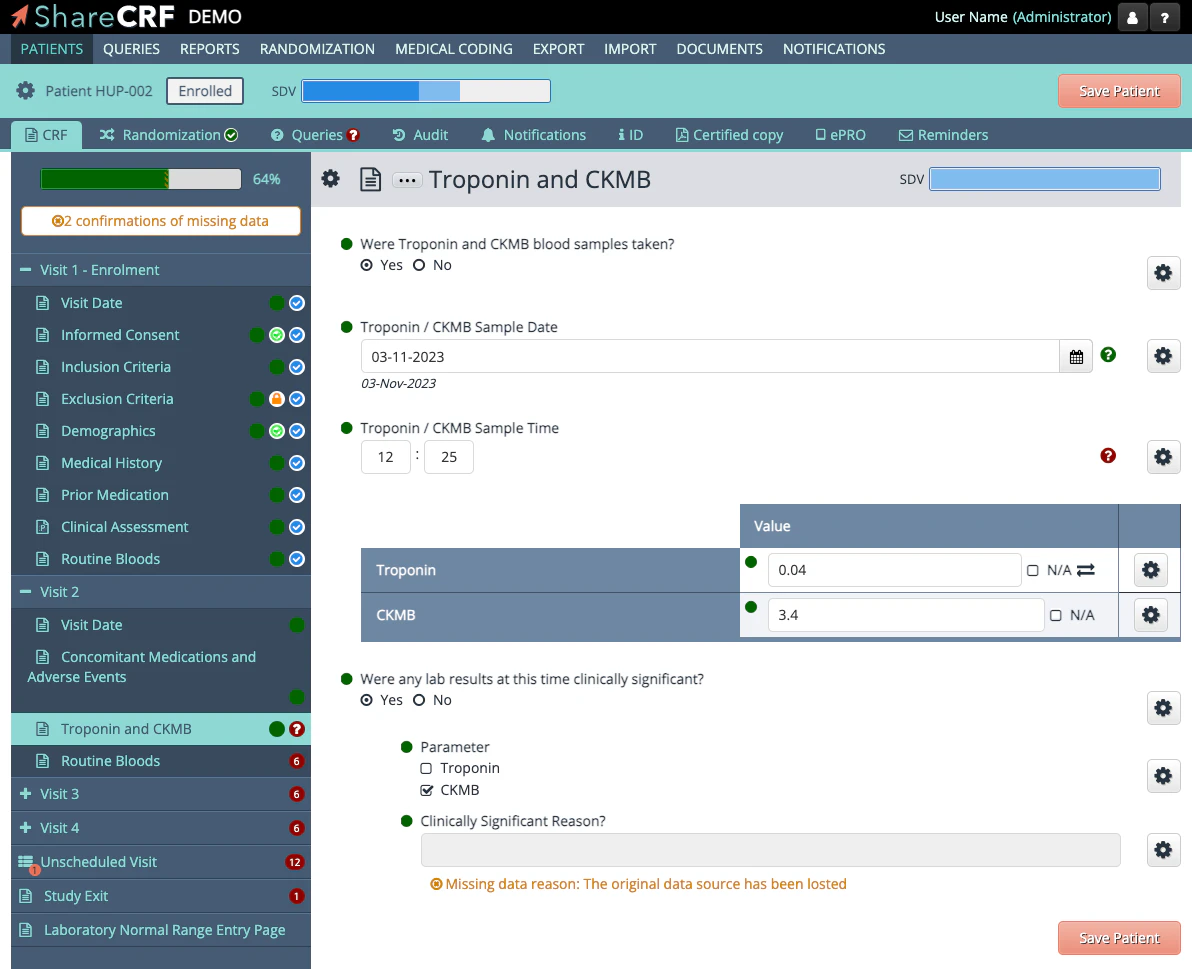
Screenshots
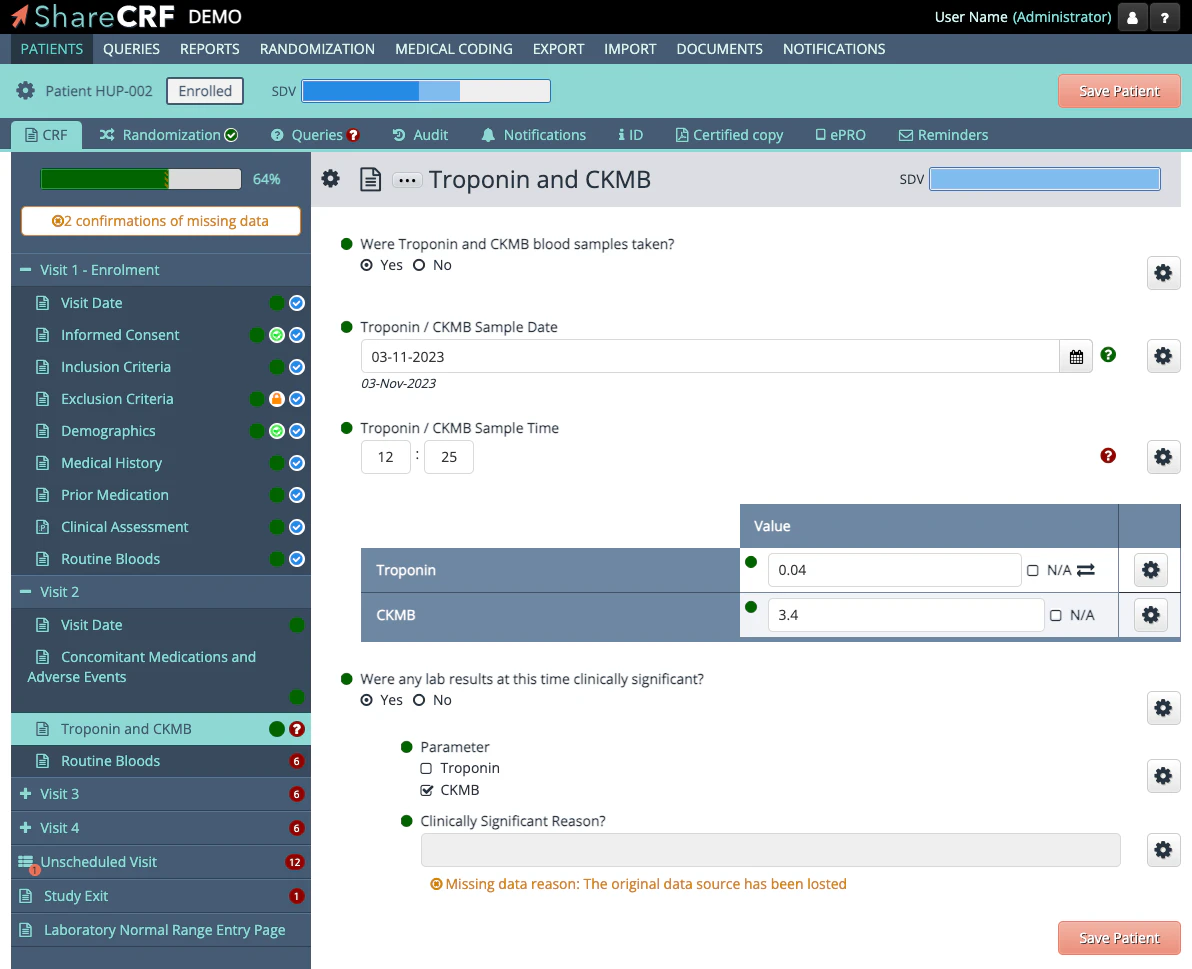

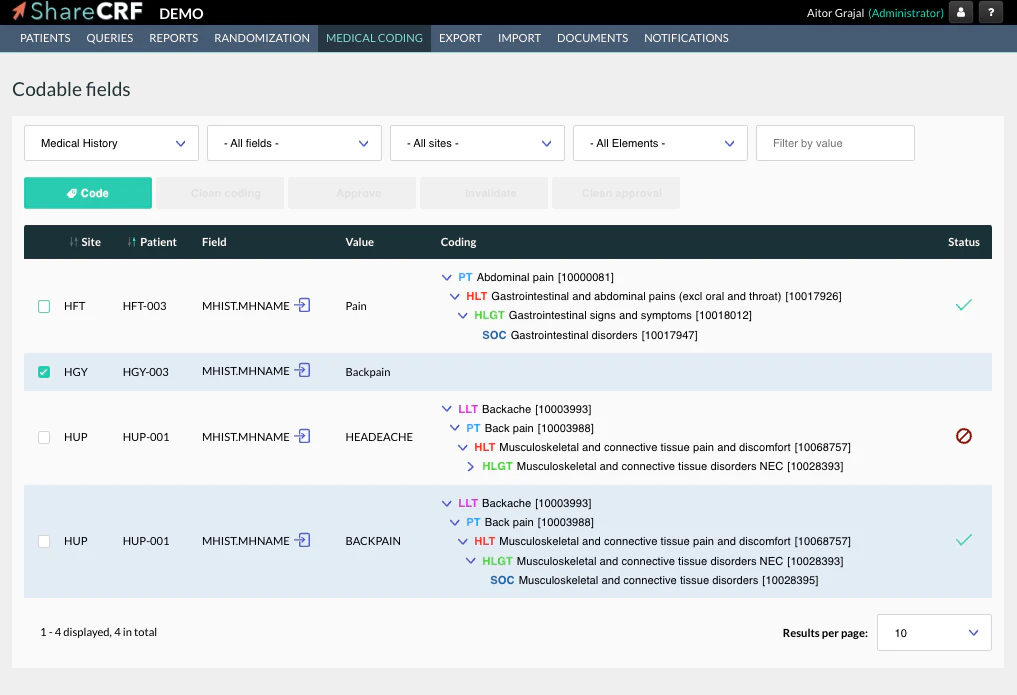
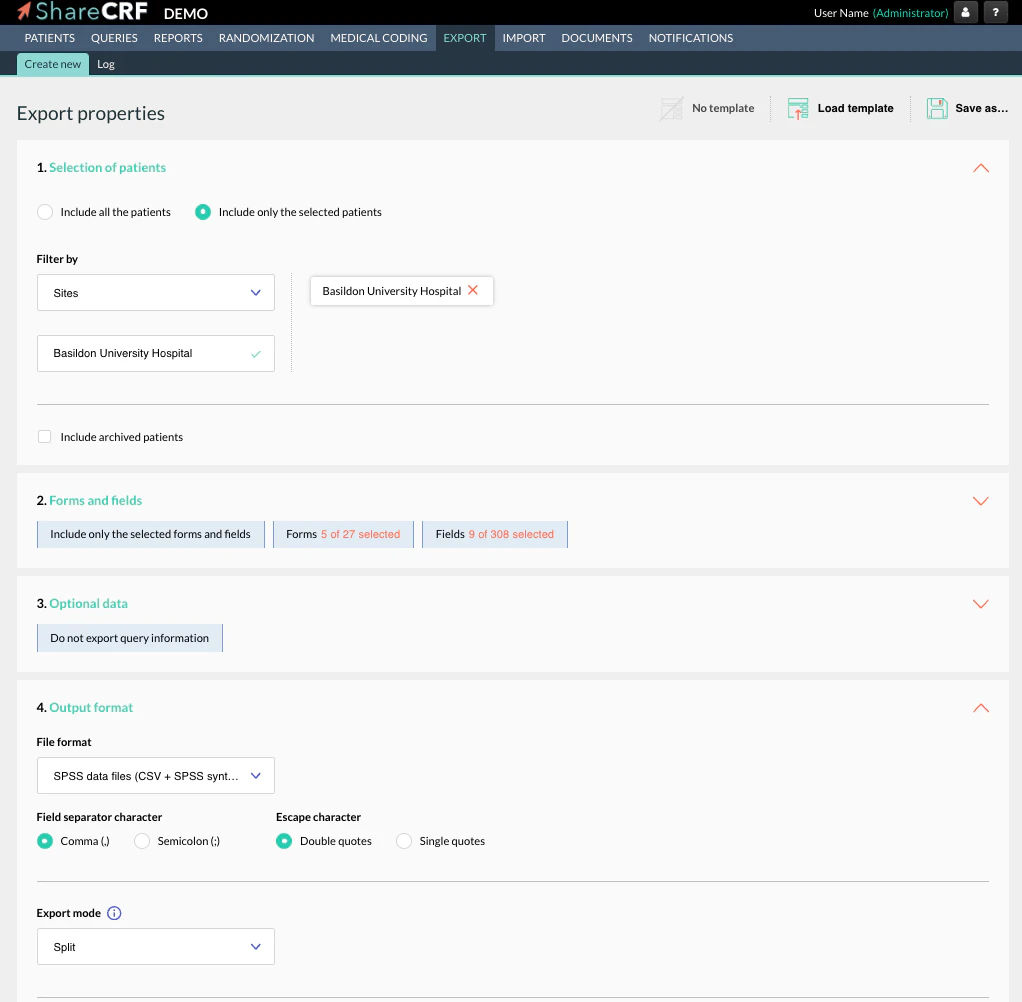
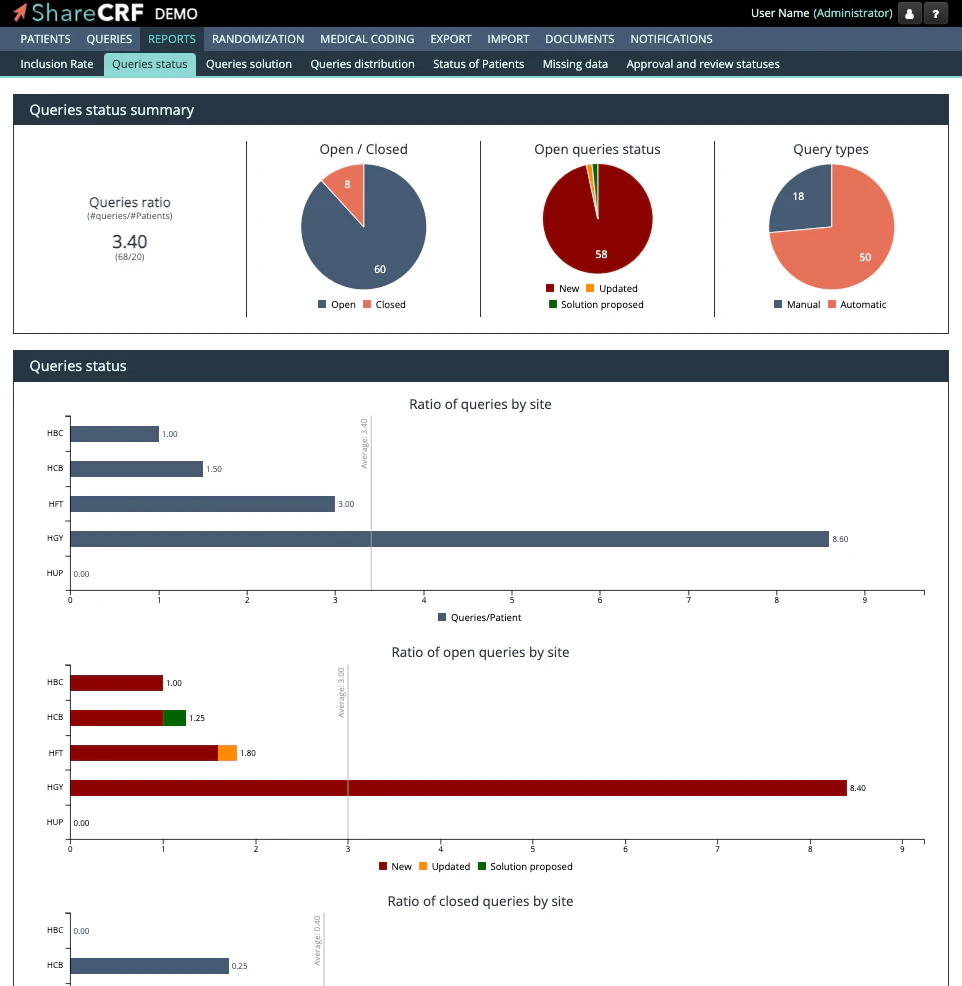
Features
FAQ
The official website of ShareCRF is https://www.sharecrf.com/
ShareCRF belongs to the Clinical Trial Management, Electronic Data Capture category.
ShareCRF offers features such as 21 CFR Part 11 Compliance, Audit Trail, Data Capture and Transfer, Data Import/Export, Document Management, Electronic Data Capture, HIPAA Compliant, Patient Monitoring, Workflow Management, Clinical Study Management, CRF Tracking, Forms Management, Remote Data Capture, Reporting/Analytics, Third-Party Integrations.