
Viedoc
Viedoc's electronic data capture (EDC) software enables users to easily collect, manage, validate and present clinical trial data.
What is Viedoc?
Screenshots


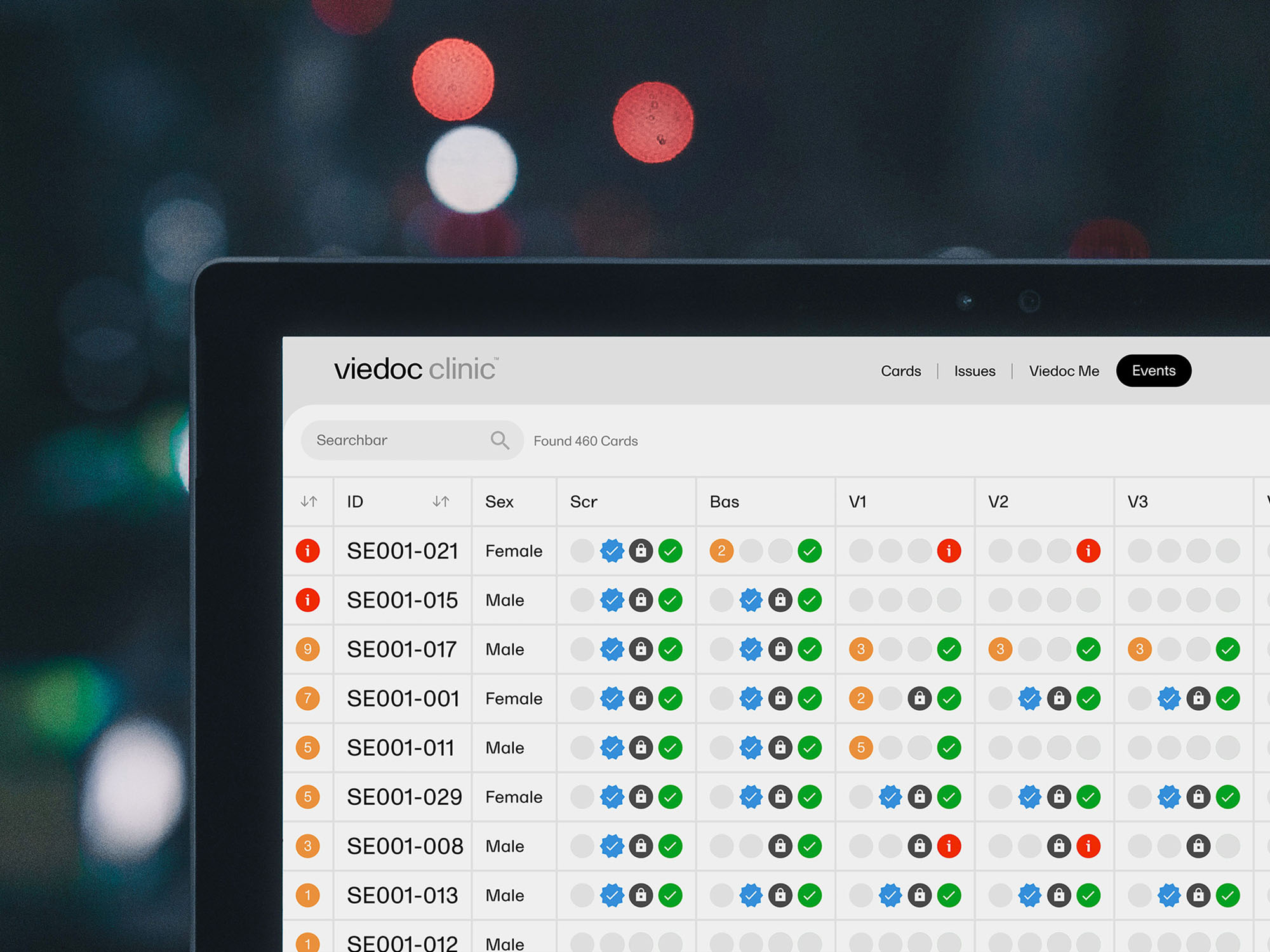
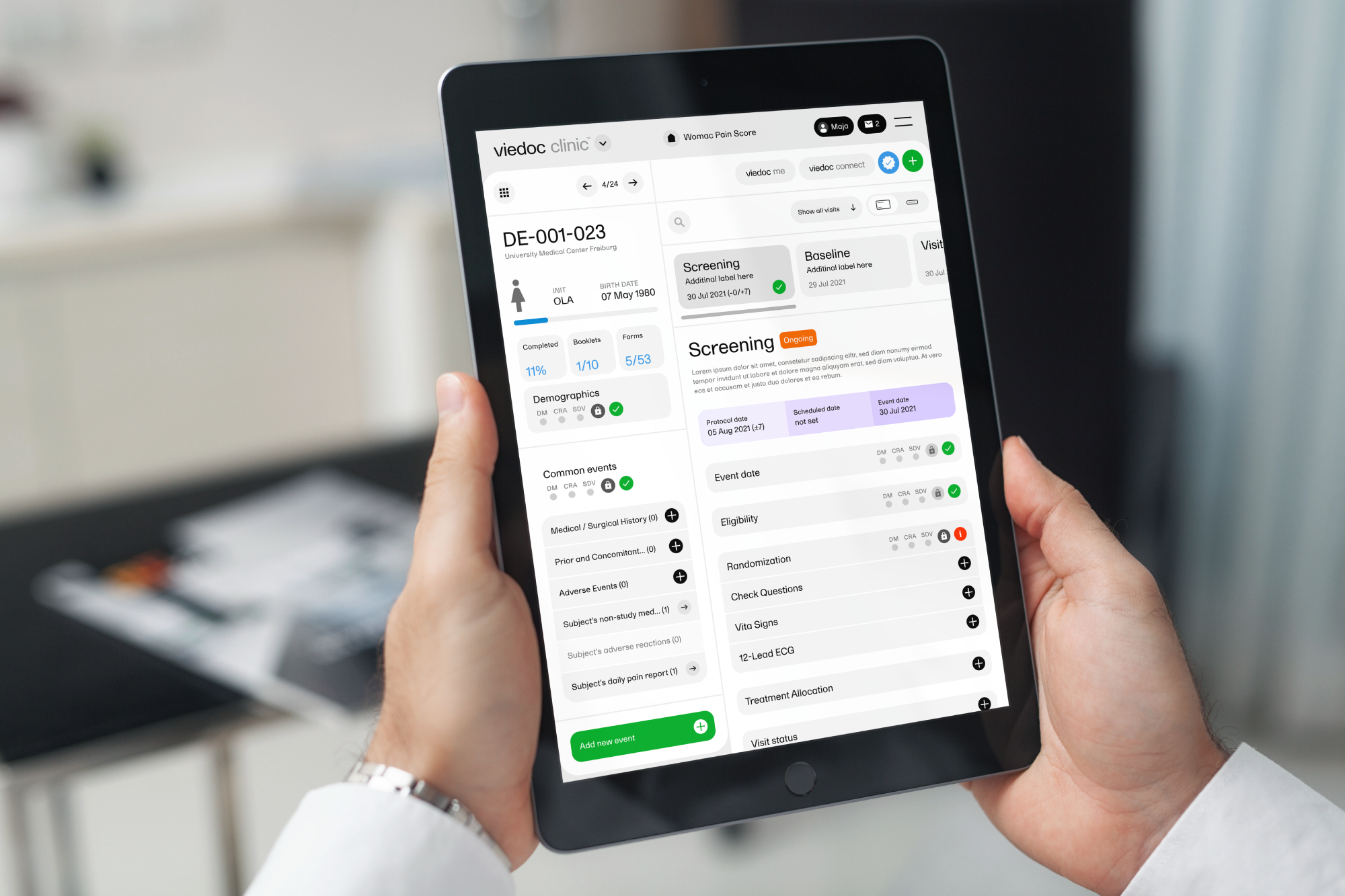
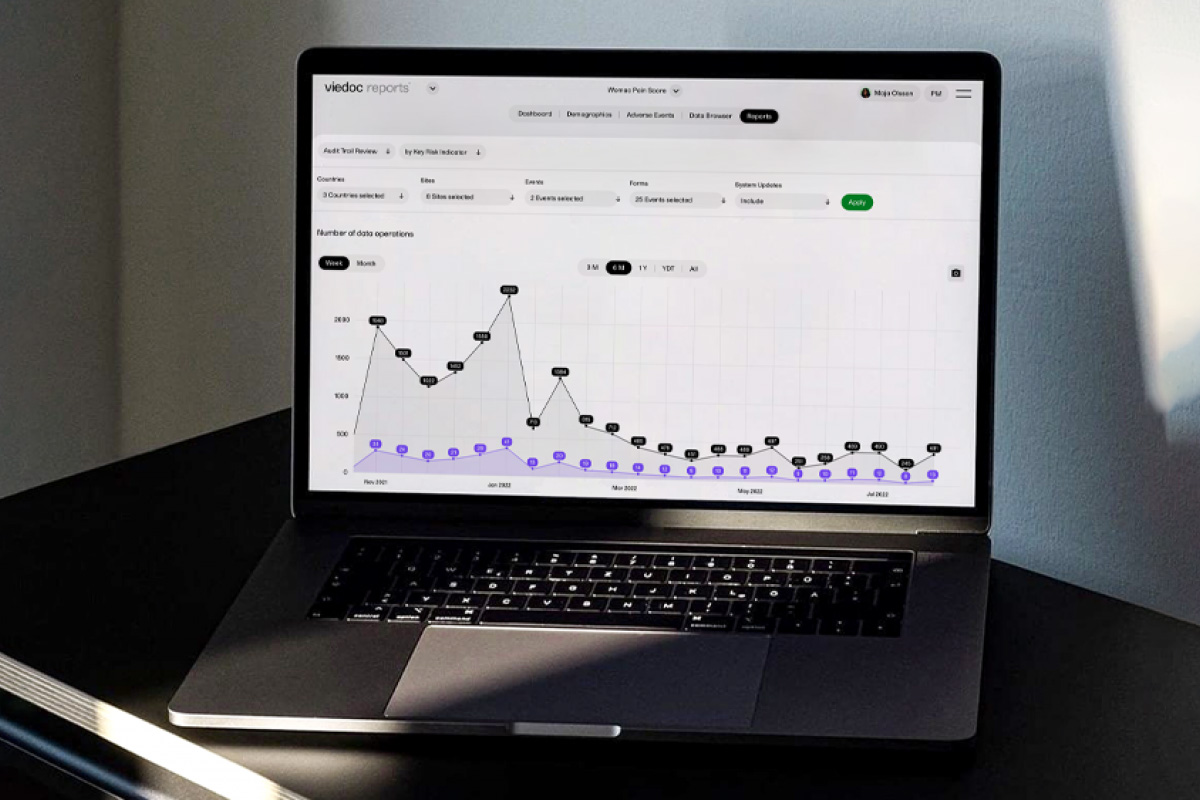

Features
FAQ
The official website of Viedoc is https://viedoc.com/
Viedoc belongs to the Clinical Trial Management, Electronic Data Capture category.
Viedoc offers features such as 21 CFR Part 11 Compliance, Audit Trail, Data Capture and Transfer, Data Import/Export, Document Management, Electronic Data Capture, HIPAA Compliant, Patient Monitoring, Workflow Management, Clinical Study Management, CRF Tracking, Forms Management, Remote Data Capture, Reporting/Analytics, Third-Party Integrations.
No, Viedoc does not offer a free trial.