
Kronus
FDA-validated clinical trial management system with document control, analytics dashboards, and electronic patient consent.
What is Kronus?
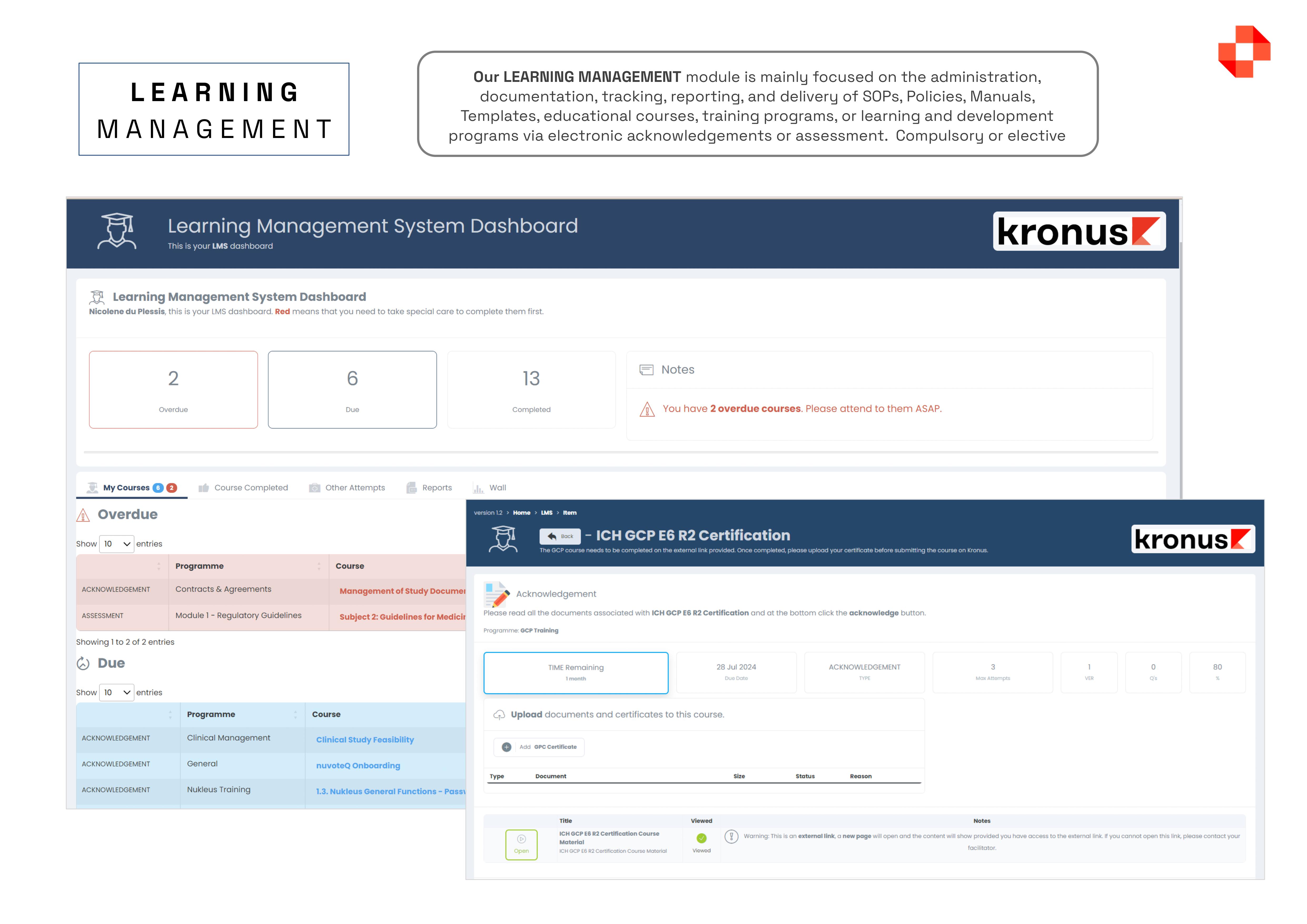
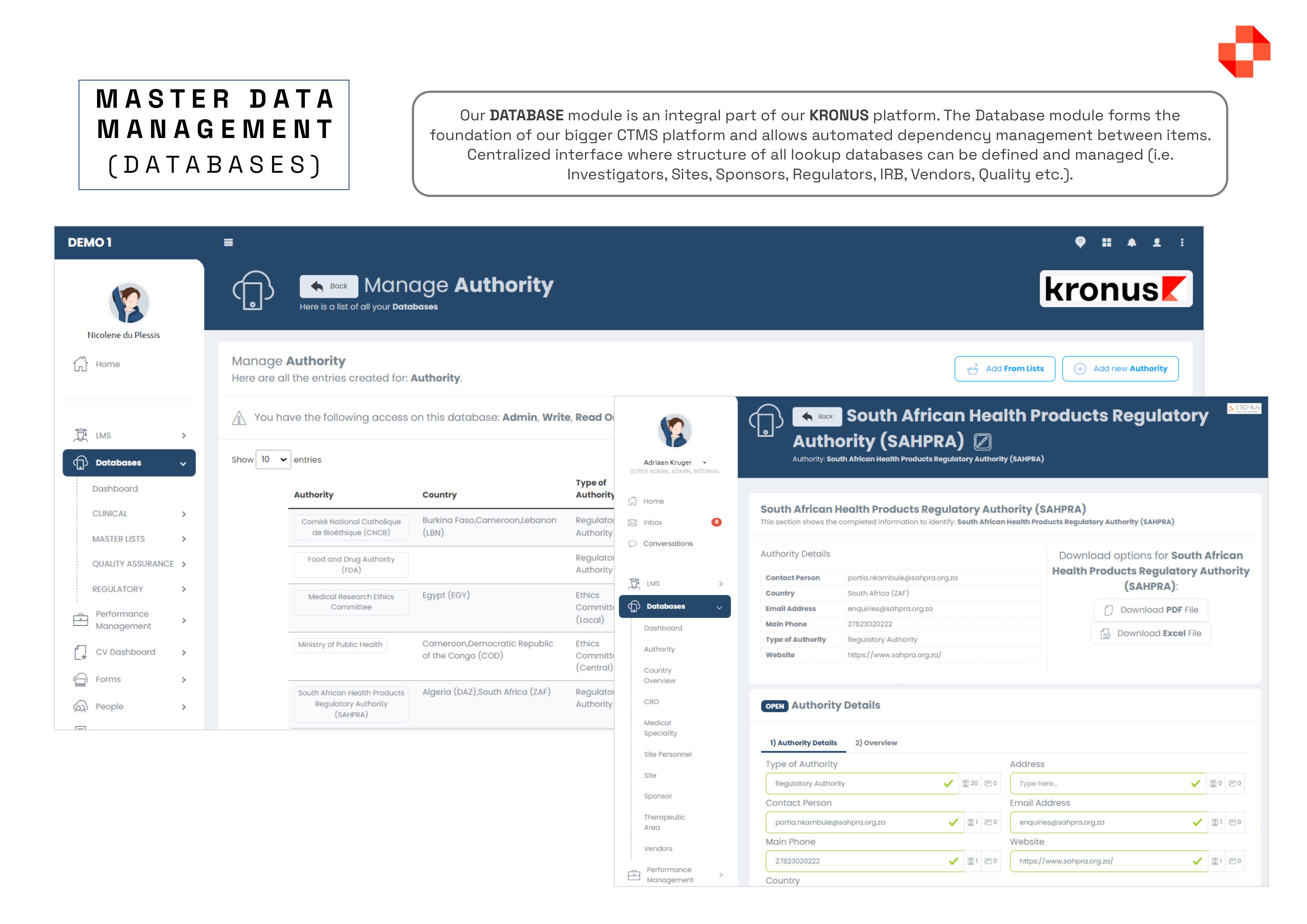
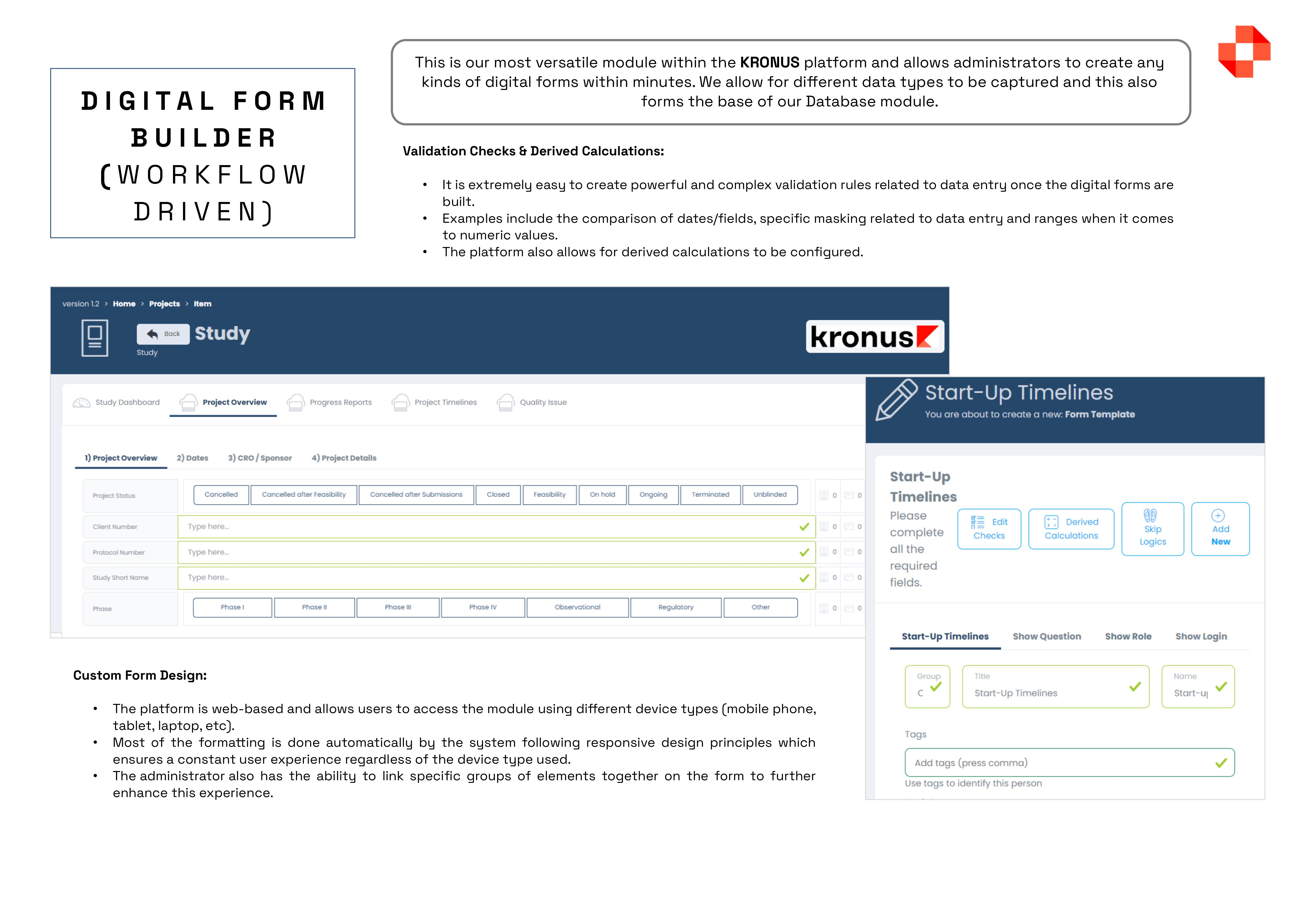
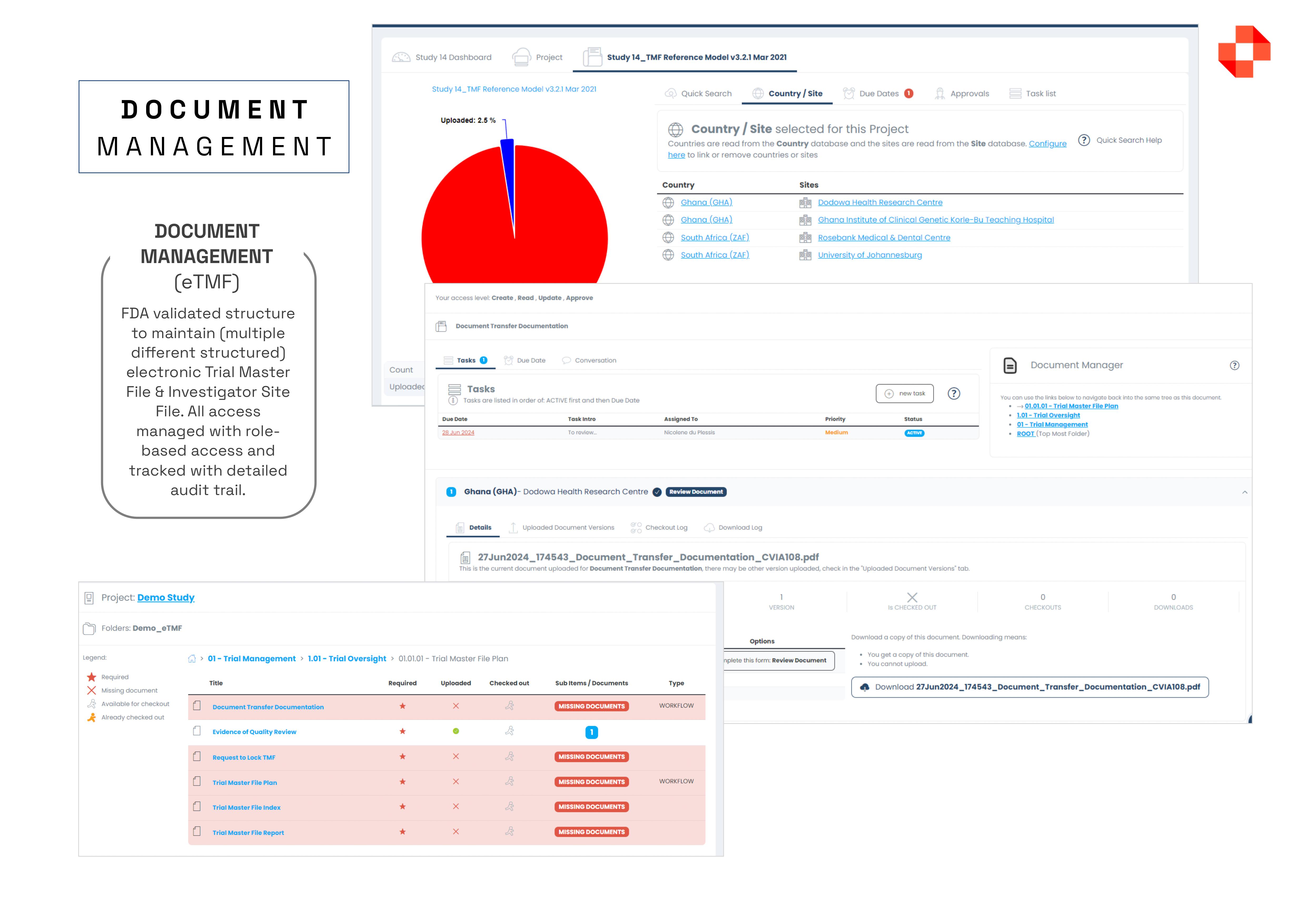
The Kronus Clinical Trial Management System (CTMS), offers a FDA-validated solution with everything you need in one package, including worksheets, documents, calendars, contacts, schedules, payments, milestones, and more, all while ensuring compliance. Some of the main features of the platform include, but are not limited to: MASTER DATA MANAGEMENT: Centralised interface for defining and managing lookup tables, such as doctors, sites, sponsors, regulators, and IRB, simplifying data management. DOCUMENT MANAGEMENT COMPLIANCE (eTMF / eISF): Maintain FDA-validated structures for Trial Master Files and Investigator Site Files, with controlled access and meticulous audit tracking. PATIENT SCHEDULING: With this module you have a single interface where you can management the entire calendar of a research centre, split across multiple subjects, multiple studies and multiple subject visits.
Screenshots


Categories
Features
21 CFR Part 11 Compliance
Audit Trail
Data Capture and Transfer
Data Import/Export
Document Management
Patient Monitoring
Workflow Management
FAQ
The official website of Kronus is https://nuvoteq.io/
The Kronus Clinical Trial Management System (CTMS), offers a FDA-validated solution with everything you need in one package, including worksheets, documents, calendars, contacts, schedules, payments, milestones, and more, all while ensuring compliance. Some of the main features of the platform include, but are not limited to: MASTER DATA MANAGEMENT: Centralised interface for defining and managing lookup tables, such as doctors, sites, sponsors, regulators, and IRB, simplifying data management. DOCUMENT MANAGEMENT COMPLIANCE (eTMF / eISF): Maintain FDA-validated structures for Trial Master Files and Investigator Site Files, with controlled access and meticulous audit tracking. PATIENT SCHEDULING: With this module you have a single interface where you can management the entire calendar of a research centre, split across multiple subjects, multiple studies and multiple subject visits.
Kronus belongs to the Clinical Trial Management category.
Kronus offers features such as 21 CFR Part 11 Compliance, Audit Trail, Data Capture and Transfer, Data Import/Export, Document Management, Patient Monitoring, Workflow Management.
No, Kronus does not offer a free trial.
Pricing
Starting at:
$1/per month
Free Trial Available